The Evolution Of Oligonucleotide Manufacturing: Engineering Considerations For Scale, Efficiency, And Facility Design
By Bill Jarvis, James Golden, and Jim Love, CRB Group

As oligonucleotide therapeutics continue to gain traction in new commercial approvals (Figure 1), manufacturers are increasingly confronting challenges that extend well beyond chemistry alone. From an engineering and facility design perspective, the question is no longer whether these therapies can be produced, but how to design robust, scalable, and safe manufacturing environments capable of supporting sustained commercial demand, particularly as large-scale indications move closer to reality.
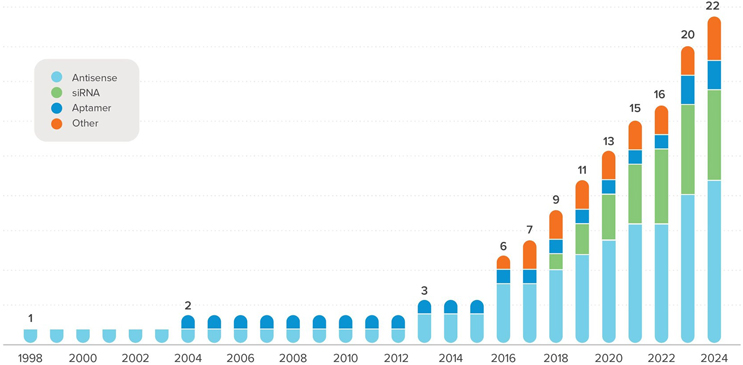
Figure 1: This growth trajectory underscores why manufacturing scale, facility footprint, and solvent management are becoming central constraints rather than downstream considerations.
From an advanced biopharmaceutical manufacturing facility design perspective, oligonucleotides represent a unique intersection of high-value chemistry, solvent-intensive processes, GMP manufacturing considerations for drugs that are largely parenteral (including some intrathecally injected molecules), and rapidly evolving production technologies. Understanding the intersection of these technologies and the impact on facility design, safety classification, manufacturing scale ceiling, and operational flexibility is critical as the industry prepares for the next generation of commercial-scale production.
Traditional Synthesis Approach Advantages
Historically, oligonucleotide manufacturing has relied on fixed bed solid-phase oligonucleotide synthesis (SPOS). In this approach, therapeutic-length oligonucleotides are assembled sequentially in a single fixed bed reactor containing a stationary synthesis resin. The initial nucleotide addition occurs at active sites on the resin, with tightly controlled reaction and wash steps following each nucleotide addition to the elongating oligonucleotide chain.
From an engineering standpoint, fixed bed reactors offer clear advantages. Near plug-flow behavior supports efficient reagent utilization and predictable washing performance. This technology is the current state of the art and is well understood by regulators and operators alike. These attributes have made fixed bed SPOS a reliable foundation for the current installed base of commercial facilities.
However, as production scales increase, the inherent limitations of fixed bed technology become more pronounced. Reactor scale is constrained by resin active site density, defined by the chemically available functional groups per unit mass of solid support, synthesis reactor column geometry, and practical considerations related to column design, including mass, lifting, ergonomics, and movement. As columns grow larger, maintaining uniform flow distribution and reaction performance becomes increasingly complex, introducing operational and safety challenges. These constraints effectively cap SPOS batch sizes at around 10 kg per batch, which prevents the technology from being a practical choice to support multi-metric ton quantities envisioned in the case of a prevalent disease commercial oligonucleotide approval.
Alternative Technologies – Stirred Tanks’ Pros And Cons
These scale limitations have driven sustained interest in alternative reactor configurations, particularly stirred slurry tank systems. In slurry-based synthesis, the resin is suspended in a well-mixed reaction vessel, thus decoupling production scale from fixed column geometry. From a facility design perspective, stirred tanks are familiar, flexible, and readily integrated into conventional manufacturing layouts. Where SPOS is limited by columns, stirred tanks can achieve much larger batch scales, often on the order of 10 times, limited only by the fabrication of the nutsche filters that capture the solid resin phase at the end of the reaction. Stirred tanks also represent an accessible equipment type that is more readily available within existing installed capacity in the world, potentially lowering barriers to entry.
Historically, one trade-off for this scalability was reduced process washing efficiency. Compared to fixed bed systems, slurry tanks exhibit lower contact efficiency between reagents and resin and require more extensive washing, driving higher solvent consumption and increased process mass intensity (PMI), or the total mass of materials required to produce a unit mass of product. For facilities designed around solvent-intensive processes, this translates directly into larger solvent storage volumes, expanded hazardous areas, and increased capital and operating costs.
Recent advances documented in patent literature have begun to close this efficiency gap. The strategic recycling of reactant and wash streams has provided an approach to significantly reduce solvent usage, potentially bringing PMI values in line with those achievable in highly optimized fixed bed processes. From an engineering standpoint, these developments directly address the aforementioned solvent-intensive facility costs, making slurry tank synthesis increasingly attractive as a tool for large-scale production of these molecules.
Alternative Technologies – Enzymatic Technology
Despite these advances, current fixed bed and slurry-based chemical synthesis approaches share a common reliance on flammable organic solvents. At commercial scale, this drives many of the most consequential facility design decisions. Hazardous occupancy classifications, fire and explosion protection systems, solvent handling infrastructure, and environmental health and safety controls all remain central design considerations that impact facility complexity and cost. Compared to non-hazardous occupancies with standard electrical classifications, classified cleanroom interior build costs in solvent-intensive facilities can represent a two- to three-times capital cost multiplier. Likewise, high alloy materials, due to inherently aggressive chemistries, can drive up raw material costs for equipment, piping, and valving on the order of five times.
Emerging enzymatic ligation technologies offer an alternative approach, synthesizing therapeutic-length oligonucleotides from shorter chemically synthesized fragments in aqueous chemistry regimes. These approaches can significantly reduce solvent use and waste generation, offering another important facility design lever.
However, upstream production of the short oligonucleotide fragments used for enzymatic ligation still currently depends on conventional chemical synthesis and thus does not eliminate the need for hazardous solvent handling in commercial facilities. By outsourcing upstream solvent processes to synthesize the fragments, an enzymatic ligation facility can cost 20% less and achieve 10 times per batch scale compared to SPOS.
Aiming to address this challenge, but preliminary in its development, is completely aqueous-based enzymatic or enzyme-assisted synthesis technology. These approaches hold the promise of eventually eliminating organic solvents from oligonucleotide production; however, they have not yet matched the flexibility of traditional SPOS chemistry, fully eliminated the need for chemically synthesized starter fragments, or addressed the separation-resolution advantages of certain downstream processes. These future facilities can combine the scale advantages discussed for stirred tanks and enzymatic synthesis and deliver facility costs of 20%-50% of SPOS. On a per kilogram basis, this means there is potential to deliver 12 to 20 times the oligo product per dollar of capital invested in support of prevalent disease indications.
Conclusion
Although the oligonucleotide field has not yet encountered a commercial therapy targeting a prevalent disease at a scale that demands substantial manufacturing expansion, it is increasingly likely that such a product will emerge. As clinical success continues to accumulate across multiple modalities, the eventual introduction of a high‑volume oligonucleotide therapy will necessitate a corresponding increase in manufacturing throughput.
Though the long-term vision for the industry may be fully enzymatic oligonucleotide synthesis and purification without reliance on chemically synthesized starting chains, the industry will move incrementally, slowed by investments in existing installed capacity, requirements for regulatory filings, and a need for technology changes to large-scale production to catch up with development and clinical-scale proof of concept. For engineering teams, this underscores the importance of designing facilities that are not only compliant and safe but also flexible enough to evolve alongside advancing process technologies. This means integrating robust hazardous material handling systems, designing for solvent recovery and recycling where feasible, and anticipating future process modifications during early facility planning. Facilities that are constrained by current assumptions may struggle to adapt as enzymatic and hybrid technologies mature.
The evolution of oligonucleotide manufacturing reflects a balance between technological innovation and engineering reality. While synthesis platforms will continue to advance, near-term commercial production will remain anchored in solvent-intensive processes. Facilities designed with flexibility, hazard mitigation, and future adaptability in mind will be best positioned to accommodate the next phase of scale.
About The Authors:
 Bill Jarvis, senior fellow – chemical and pharmaceutical process, CRB Group, has more than 38 years of broad-based chemical engineering experience. His experience ranges from exploratory process development through lab and pilot plant scale-up, process design, process start-up, and process optimization. Jarvis specializes in API production facility design, oral solid dose formulation (OSD) processes, plasma-derived therapy products, and oligonucleotide and peptide facility design. Jarvis also helped develop the International Society for Pharmaceutical Engineering’s Community of Practice (CoP) concept, serving as a past chair of the API CoP, and contributing to the containment and OSD CoPs.
Bill Jarvis, senior fellow – chemical and pharmaceutical process, CRB Group, has more than 38 years of broad-based chemical engineering experience. His experience ranges from exploratory process development through lab and pilot plant scale-up, process design, process start-up, and process optimization. Jarvis specializes in API production facility design, oral solid dose formulation (OSD) processes, plasma-derived therapy products, and oligonucleotide and peptide facility design. Jarvis also helped develop the International Society for Pharmaceutical Engineering’s Community of Practice (CoP) concept, serving as a past chair of the API CoP, and contributing to the containment and OSD CoPs.
 James Golden, fellow – chemical process, CRB Group, has more than 20 years of experience as a process engineer with broad experience in hazardous chemical processing and handling; API processes; oral solid dose processing including hazardous containment design; process utility systems design such as purified water, clean steam, and clean compressed gases; and, most recently, oligonucleotide facility design. Golden has extensive experience with code implications to process and facility design for the National Fire Protection Association (NFPA), International Code Council (ICC), and other regulatory and safety compliance bodies. Golden has also been a member of the API Community of Practice Committee for the International Society of Pharmaceutical Engineers (ISPE).
James Golden, fellow – chemical process, CRB Group, has more than 20 years of experience as a process engineer with broad experience in hazardous chemical processing and handling; API processes; oral solid dose processing including hazardous containment design; process utility systems design such as purified water, clean steam, and clean compressed gases; and, most recently, oligonucleotide facility design. Golden has extensive experience with code implications to process and facility design for the National Fire Protection Association (NFPA), International Code Council (ICC), and other regulatory and safety compliance bodies. Golden has also been a member of the API Community of Practice Committee for the International Society of Pharmaceutical Engineers (ISPE).
 Jim Love, oligonucleotide/peptide market leader, CRB Group, has comprehensive experience in strategy development, planning and analysis, capital project management, and engineering design. He has led many types of projects, ranging from project chartering and site selection to design and construction execution strategy development and implementation to tactical facility design. His broader professional experience includes biotechnology, oligo/peptide, industrial extraction processes, and oral solid dose technologies.
Jim Love, oligonucleotide/peptide market leader, CRB Group, has comprehensive experience in strategy development, planning and analysis, capital project management, and engineering design. He has led many types of projects, ranging from project chartering and site selection to design and construction execution strategy development and implementation to tactical facility design. His broader professional experience includes biotechnology, oligo/peptide, industrial extraction processes, and oral solid dose technologies.
