Suppressor tRNA Therapeutics: A New Frontier In Treating Genetic Disease
By Elisabeth Gardiner, Ph.D., chief scientific officer, Tevard Biosciences, Inc.
RNA therapeutics have fundamentally reshaped the landscape of modern medicine. From mRNA vaccines to siRNAs and antisense oligonucleotides (ASOs), each breakthrough has expanded our capacity to intervene at the genetic and molecular level. Yet despite these remarkable advances, a critical gap remains and millions of patients with diseases caused by nonsense mutations have been left without personalized treatment options.
Nonsense mutations, which introduce premature termination codons (PTCs) that halt protein translation prematurely, represent an intractable challenge for genetic medicine. Existing gene therapy solutions may be constrained by payload size, mutation specificity, and scalability. PTCs lead to pathological proteins that rob patients of healthy lives and account for 10% to 15% of all inherited genetic diseases. Nonsense mediated diseases include devastating conditions such as Duchenne muscular dystrophy (DMD), genetic cardiomyopathies, and developmental and epileptic encephalopathies (DEEs). No approved therapies broadly target nonsense mutations across these conditions. The unmet medical need is enormous.
Suppressor tRNAs offer a compelling answer. This emerging therapeutic modality is engineered to read through premature stop codons and restore the production of full-length, functional protein. With the potential to address millions of patients across multiple diseases with a single scalable platform, suppressor tRNAs are a game-changing advancement for genetic medicine.
Nonsense Mutations: Why Existing Therapies Fall Short
In healthy cells, tRNAs decode codons on mRNA and ferry amino acids to the ribosome, assembling proteins one building block at a time until a natural stop codon signals completion. In patients with nonsense mutations, this process is hijacked: a PTC triggers premature termination, yielding a truncated protein incapable of performing its essential function.
Traditional gene therapy cannot solve this problem. Payload constraints make it challenging to deliver large disease-causing genes, such as dystrophin in DMD or titin in dilated cardiomyopathy, into viral and non-viral vectors. Conventional mutation-specific therapies require bespoke development programs, making them economically and logistically untenable.
Suppressor tRNAs As A Novel Therapeutic Modality
Engineered suppressor tRNAs are purpose-built to address the issue of premature stop codons. By recognizing PTCs and inserting the appropriate amino acid to allow translation to continue, they restore the production of full-length, endogenous protein. This matters clinically as functional restoration is the ultimate goal of genetic medicine.
An analysis of 16,000 pathogenic nonsense mutations revealed that all nonsense codons arise from just 19 unique point mutations. Arg-TGA and Gln-TAG alone account for nearly half of all cases genome-wide. This remarkable convergence means that a small number of suppressor tRNAs could, in principle, address the vast majority of nonsense mutation-driven genetic diseases.
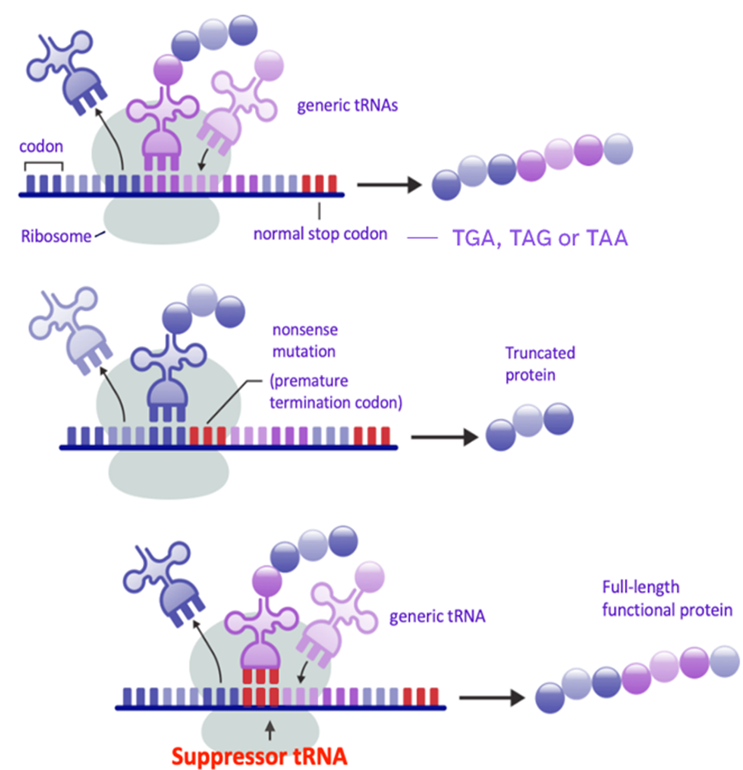
Suppressor tRNA Technology
Suppressor tRNAs offer a distinctive set of advantages that set them apart from other genetic medicine platforms. Their small molecular footprint removes the payload constraints that have long limited traditional gene therapy, enabling targeting of even the largest genes in the human body. Their portability means a single therapeutic product could potentially treat a broad spectrum of diseases sharing the same nonsense mutation, a radical departure from the one-drug-per-disease paradigm. Precise targeting ensures that only proteins arising from mutated transcripts are affected. Optimized vector design, promoter sequences, and capsid selection facilitate safe and tissue-specific delivery. And because the approach requires only a single low-dose AAV administration, it offers unique durability. Suppressor tRNAs work at the level of the ribosome to rescue translation, restoring the body’s own protein-making machinery without genomic modifications.
Compelling Preclinical Results Across Muscle And Cardiac Disease
At the OPT Congress 2026, Tevard presented preclinical data that underscores the transformative potential of this platform. Engineered suppressor tRNAs demonstrated greater than 80-fold gains in readthrough activity, while maintaining exquisite PTC specificity, and critically, without inducing unintended readthrough of native stop codons. These results establish a new performance benchmark for the field.
In DMD, nonsense mutations affecting dystrophin account for roughly 15% of cases, approximately 4,000 patients who are largely unreachable by current therapies, including exon-skipping approaches. In disease relevant mouse models, Tevard’s suppressor tRNA restored full-length dystrophin protein and drove significant recovery of motor function and muscle performance at 12 weeks post-dosing, approaching levels seen in wild-type mice. Notably, both TAA and TGA suppressor tRNAs demonstrated comparable efficacy in rescuing dystrophin expression, a finding that speaks to the platform’s versatility.
The results in dilated cardiomyopathy caused by TTN mutations (DCM-TTN) are equally striking. With more than 100,000 patients across the U.S. and EU estimated to carry TTN nonsense mutations, this disease represents a massive area of unmet need. In mouse models, a single intravenous dose of suppressor tRNA achieved dose-dependent and durable restoration of full-length titin protein. Critically, titin rescue was accompanied by normalization of proteomic homeostatic signatures, suggesting a broad correction of disease biology rather than a surface-level effect. The translational relevance of these findings is further supported by data in human iPSC-derived cardiomyocytes carrying TTN mutations. Treatment with suppressor tRNAs produced dose-dependent recovery of full-length titin protein, with both TGA and TAA variants demonstrating efficacy, reinforcing the platform’s potential to translate from animal models to human disease.
One Therapy, Many Diseases: Redefining The Development Model
The predictability of nonsense mutations, converging on just 19 unique point mutations across thousands of diseases, is not merely a scientific curiosity. It is the foundation of a fundamentally different development strategy. Unlike traditional gene therapy, suppressor tRNAs make a disease-agnostic model possible: one well-engineered therapy targeting a specific PTC could address patients across an entire spectrum of disease. Rather than mounting a separate clinical trial for each patient, suppressor tRNAs unlock a model for testing a single therapeutic across multiple related conditions that share the same nonsense mutation. The result is greater efficiency for drug development and the ability to reach patients that have long been underserved. Suppressor tRNAs are not simply a new tool in the RNA toolbox. This technology represents an opportunity to rethink what genetic medicine can do.
About The Author:
 Elisabeth Gardiner, Ph.D., is chief scientific officer at Tevard Biosciences, spearheading the company’s pioneering tRNA-based gene therapies for rare diseases. With over 25 years in the biopharmaceutical industry, she has directed and managed drug discovery and development efforts yielding 11 IND filings, four Phase 1/2 trial candidates, and one Phase 3 candidate.
Elisabeth Gardiner, Ph.D., is chief scientific officer at Tevard Biosciences, spearheading the company’s pioneering tRNA-based gene therapies for rare diseases. With over 25 years in the biopharmaceutical industry, she has directed and managed drug discovery and development efforts yielding 11 IND filings, four Phase 1/2 trial candidates, and one Phase 3 candidate.
Prior to Tevard, Gardiner held senior R&D leadership roles at Tactile Therapeutics, Alterome Therapeutics, Aravive, and Kinnate Biopharma, where she directed multidisciplinary teams and advanced programs from discovery to clinical-stage development across neurology, oncology, and rare disease. Gardiner gained experience in tRNA biology while working at aTyr Pharma and at Scripps Research Institute in the lab of Paul Schimmel.
Gardiner’s commitment to the ethical development of effective and accessible medicines is her key focus in life. In addition to her professional work, she is a patient advocate in the rare disease and oncology space.
Gardiner earned her Ph.D. from the University of Wisconsin-Madison and holds a bachelor’s and master’s degree from Texas A&M University.
