Bridging The Manufacturing Chemistries Between Drug Research And Development For Morpholino Oligos
By Yongfu Li, Ph.D., Gene Tools, LLC

Phosphorodiamidate Morpholino oligonucleotides (PMO or Morpholino) are increasingly recognized as a distinct class of therapeutic antisense agents for a variety of genetic diseases. However, variations in synthetic methodologies among manufacturers lead to inconsistencies in oligonucleotide structural entities, primarily due to differing linker strategies. These inconsistences in structural characterization can complicate development efforts.
Recent guidelines1 from the FDA indicate that the "platform nature" of oligonucleotides — where antisense oligonucleotides share similar building blocks — may enable regulatory efficiencies. The FDA can thus expedite approval of new oligonucleotide drugs that share structural features and chemical modifications to previously approved drugs by leveraging existing clinical pharmacology data, established safety profiles, and prior regulatory pathways for similar molecular platforms.
Structural similarity can speed approval in the following aspects:
- Platform Technology Designation: The FDA introduced a Platform Technology Designation Program (May 2024) to streamline the approval of therapies using shared technology platforms, which could apply to platform-based oligonucleotides.
- Predictable Safety and Pharmacokinetics: Similar structural architectures often result in comparable pharmacokinetic behaviors (absorption, distribution, metabolism, and excretion), reducing the burden of de novo nonclinical studies.
- Prior Examples: Several recent oligonucleotide approvals have mimicked the chemical structure and delivery method of earlier successful compounds to target new sequences.
- Streamlined CMC: For synthetic oligonucleotides with highly similar primary structures, the FDA may focus more on analytical sequence verification rather than extensive new potency bioassays.
Therefore, establishing a well-defined and optimized structural platform for the Morpholino field is highly desirable to support streamlined drug development and enable more efficient regulatory approval.
Current Structural Types Of Morpholino Oligos
Currently, there are three 5’-end structural options for Morpholino synthesis (Figure 1).
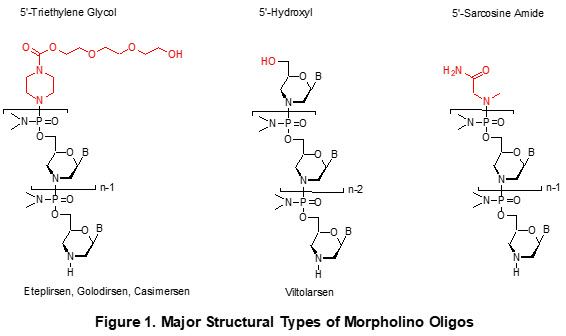
Sarepta Therapeutics synthesizes PMOs with triethylene glycol at the 5’-end. So far, we have three FDA-approved drugs (eteplirsen,2 golodirsen,3 and casimersen4) containing this 5’-end chemistry. Nippon Shinyaku’s Morpholino drug (viltolarsen5) contains a hydroxyl at the 5’-end. These drugs are approved to treat Duchenne muscular dystrophy (DMD), a severe muscle wasting disease caused by a disruption of the dystrophin mRNA reading frame, resulting in an out-of-frame transcript and a nonfunctional or absent dystrophin protein. These unmodified Morpholinos have poor intracellular penetration capabilities, resulting in marginal therapeutic benefit. Plausibly, these drugs can initially enter leaky muscle cells, but further delivery of the Morpholino oligo into treated muscle cells has been hampered until the muscles degenerate to become leaky again. The cycles of improvement and degeneration compromise the therapeutic outcome.6
Generally, diseases that do not create a “leaky cell” scenario would be even more difficult to treat with an unmodified Morpholino. To overcome this delivery hurdle and enhance therapeutic efficiency, modifying the oligos to improve in vivo delivery is crucial. However, structures such as viltolarsen lack a functional site at the 5’-end for structural modification, limiting the ability to incorporate features such as in vivo delivery components.
The manufacturing process developed by Gene Tools employs sarcosine as a linker on solid-phase support. The amino group of sarcosine provides a reactive site at the 5’-end, enabling oligonucleotide elongation or the installation of functional groups prior to oligo assembly.7 Installation of functional groups at the oligo’s 5’-end increases the range of possible modifications.8 Additionally, 3’-end modifications are achievable using the secondary amine of the terminal Morpholino subunit.
Modifying one or both ends of a Morpholino oligo enables functionalization, particularly for in vivo delivery applications. A promising approach involves antibody conjugation to facilitate targeted delivery.9 Another popular approach involves conjugating cell-penetrating peptides to the Morpholino oligo, enhancing cellular uptake.10 Gene Tools developed Vivo-Morpholinos, which incorporate an octa-guanidine dendrimer coupled with the Morpholino oligo.11 Vivo-Morpholinos demonstrate high efficiency when delivered systemically via intravenous (IV) injection. They can also be administered locally by intraperitoneal (IP) injection or by direct injection to the area of interest.12 The invention of Vivo-Morpholino has encouraged discovery and development at different research stages.13
Incremental improvements over the past decades using sarcosine linker chemistry have allowed routine synthesis of high-quality Morpholino oligos with a variety of modifications and functionalities. However, there are still two critical problems to be resolved (Figure 2):
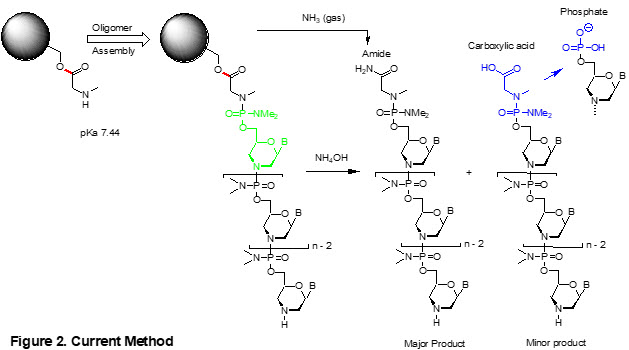
(1) Weak base (pKa 7.44) of the amino group of sarcosine: This amino group is not nucleophilic enough to achieve high-yield coupling with the first Morpholino subunit (in green in Figure 2), leading to a significant amount of N-1 side products. A more nucleophilic amine is needed to minimize these side products.
(2) The ester linkage of sarcosine with solid phase support: Post oligo assembly, aqueous ammonia cleaves the ester linkage (in red in Figure 2) to produce sarcosine amide as a predominant product. However, the water in aqueous ammonia can also hydrolyze the ester to produce carboxylic acid (in blue in Figure 2). The carboxylic acid is only one Dalton larger in molecular weight than the amide, making it very difficult to detect by mass spectroscopic analysis and remove by simple purification methods. Furthermore, the carboxylic acid degrades to form a phosphate compound (in blue in Figure 2), adding one more impurity to the product profile. The use of gaseous ammonia can prevent carboxylic acids. However, for large-scale synthesis, achieving adequate penetration of ammonia throughout a large oligo cake presents a significant practical challenge.
Improved Structural Type Of Morpholino Oligo
Recently, a novel method that uses morpholin-2-ylmethanol (morpholinol) as a linker has been developed (Figure 3).14 This new amino group of morpholinol (pKa 8.36) is about 10 times more nucleophilic than sarcosine, allowing for more complete coupling with the first Morpholino subunit, thus improving the oligo quality by reducing the amount of N-1 side products.
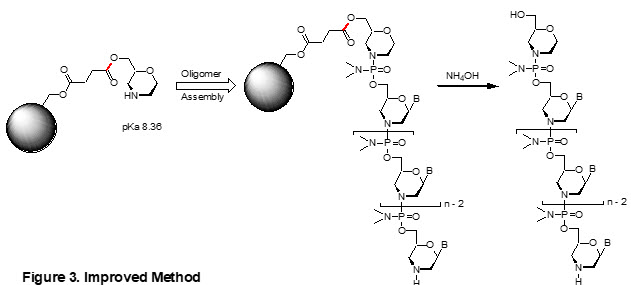
More importantly, the ester linkage (in red in Figure 3) to the solid phase support was oriented the other way than that of sarcosine. Following oligo assembly, cleavage of the ester linkage by aqueous ammonia, regardless of ammonolysis or hydrolysis, consistently yields a hydroxyl at the oligo’s 5’-end. This new linker structure prevents the formation of carboxylic acid and its subsequent phosphate side product observed with the sarcosine linker.
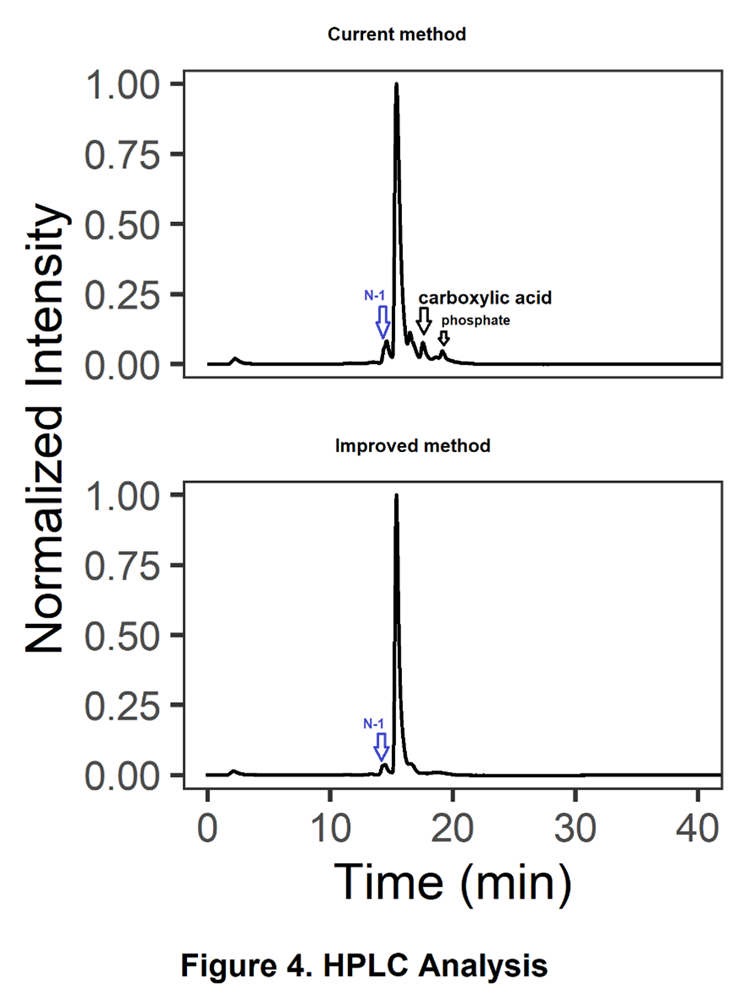
We compared two oligos with identical sequences, one synthesized using the sarcosine linker (current method) and the other using the morpholinol linker (improved method). HPLC analysis of the crude samples (Figure 4) demonstrated that the improved method: (a) reduced N-1 deletion, and (b) eliminated the carboxylic acid side product and its subsequent phosphate derivative. This improvement enables the production of Morpholino oligos with enhanced quality and higher purity.
Narrowing Down The Oligo Structural Platform
According to the definition used by the FDA, the three Morpholino oligo structural variants shown in Figure 1 are classified as distinct platforms due to differences in linker chemistry. However, in-house analysis confirmed that these three structurally distinct versions of the same oligo sequence exhibit the same biological activity.
Morpholino oligos made by this improved method (Figure 3) emerge as a new structural type. Again, when compared with the oligo made from the sarcosine linker, the oligo produced with the morpholinol linker retains biological activity, suggesting that linker architecture does not significantly influence. Since the different linkers exhibit identical biological activity, linker variation becomes a potential regulatory consideration. Since the FDA considers each structural type as a separate platform for approval purposes, increasing diversity in linker strategies across manufacturers could further complicate the regulatory landscape for Morpholino oligo development.
Morpholino oligos have tremendous therapeutic potential for treating a variety of genetic diseases. To support efficient and cost-effective advancement of Morpholino drug development, coordinating Morpholino chemical structural consistency is highly desirable. Reducing the number of structural variants — ideally converging toward a single standardized structural framework — could help streamline regulatory pathways. Within such a unified structural platform, new Morpholino oligos differing primarily in sequence could potentially be evaluated more efficiently, enabling faster regulatory review and approval.
We do not encourage the creation of innumerable structural variants by new manufacturers. Instead, we advocate for the morpholinol structural type (Figure 3) as the Morpholino platform standard, as it consistently produces oligos of the highest quality to date. In addition, the architectural structure is distinctly unique. The uniqueness lies in the fact that the entire oligo is composed of Morpholino subunits, including the morpholinol linker that functions as an abasic subunit. The abasic subunit will not bind to genetic targets and thus does not contribute to any biological activity, but it allows 5’-end functionalization of the oligo. Its amino group acts analogously as the amino group of sarcosine. The extensive chemistries developed using the sarcosine linker can be easily adapted for oligo assembly and modification within this morpholinol framework.
Summary
Phosphorodiamidate Morpholino oligonucleotides are a unique class of antisense therapeutics for the treatment of genetic diseases. However, variability in synthetic methodologies among manufacturers can result in inconsistent structural entities, potentially complicating regulatory approval for drug development. We present an optimized synthetic process for Morpholino oligonucleotide production and advocate the importance of a standardized structural platform to facilitate efficient translation from preclinical development to clinical-scale manufacturing in the Morpholino therapeutic industry.
References:
- U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER), Center for Biologics Evaluation and Research (CBER). Platform Technology Designation Program for Drug Development Guidance for Industry. FDA Guidance Document. May 2024.
- Lim KRQ, Maruyama R, Yokota T. Eteplirsen in the Treatment of Duchenne Muscular Dystrophy. Drug Des Devel Ther. 2017; 11:533–545.
- Frank DE, Schnell FJ, Akana C, et al. Increased Dystrophin Production with Golodirsen in Patients with Duchenne Muscular Dystrophy. Neurology. 2020; 94(21):e2270–e2282.
- Wagner KR, Kuntz NL, Koenig E., et al. Safety, Tolerability, and Pharmacokinetics of Casimersen in Patients with Duchenne Muscular Dystrophy Amenable to Exon 45 Skipping: A Randomized, Double-blind, Placebo-controlled, Dose-titration Trial. Muscle Nerve. 2021;64(3):285-292.
- Roshmi RR, Yokota T. Pharmacological Profile of Viltolarsen for the Treatment of Duchenne Muscular Dystrophy: A Japanese Experience. Clin Pharmacol. 2021;13:235-242.
- Li YF. To Discover, Develop and Deliver a Right Drug: A Showcase for Antisense Technology. J Drug Discov Develop and Deliv. 2014;1(1): 3.
- Li YF. Functionalizing Morpholino Oligos for Antisense Drug Research and Development. J Drug Discov, Develop and Deliv. 2016;3(1):1021.
- Li YF. End-Modifications on Morpholino Oligos. Methods in Molecular Biology, Volume 1565: Morpholino Oligomers: Methods and Protocols. Humana Press (Springer). 2017; Chapter 4: 39-50.
- Cochran M, Marks I, Albin T., et al. Structure-Activity Relationship of Antibody-Oligonucleotide Conjugates: Evaluating Bioconjugation Strategies for Antibody-Phosphorodiamidate Morpholino Oligomer Conjugates for Drug Development. J Med Chem. 2024;67(17):14868-14884.
- Leckie J, Yokota T. Potential of Cell-Penetrating Peptide-Conjugated Antisense Oligonucleotides for the Treatment of SMA. Molecules. 2024; 29:2658.
- Li YF, Morcos PA. Design and Synthesis of Dendritic Molecular Transporter that Achieves Efficient in Vivo Delivery of Morpholino Antisense Oligo. Bioconjugate Chem. 2008; 19:1464–1470.
- Morcos PA, Li YF, Jiang S. Vivo-Morpholinos: A Non-Peptide Transporter Delivers Morpholinos into a Wide Array of Mouse Tissues. BioTechniques. 2008; 45(6):613-623.
- Li YF. In Vivo Delivery of Morpholino Oligos as Therapeutics: What Barriers Still Exist? J Drug Discov Develop and Deliv. 2016; 3(1): 1017.
- Li YF, Summerton DB. Morpholino Oligomer Synthesis. 2026; US19/545,920.
- Reeves, MD, Weller DD, Li YF. Synthesis of Morpholino Oligomers Using Doubly Protected Guanine Morpholino Subunits. 2011; US8076476.
- Li YF. Molecular Transporter Compositions Comprising Dendrimeric Oligoguanidine with a Triazine Core that Facilitate Delivery into Cells in vivo. 2011; US7935816.
About The Author:
 Yongfu Li, Ph.D., obtained his doctorate from the University of Vienna, Austria. After doing postdoctoral research work at the University of Cambridge, U.K., Li joined AVI Biopharma, Inc., USA (now Sarepta Therapeutics, Inc.). Four years later, he became head chemist at Gene Tools, LLC, and has been serving in that role ever since. For nearly 30 years, Li has gained extensive experience guiding structural modifications and Morpholino oligo functionalization. His most remarkable contributions to Morpholino chemistry have included the invention of double-protection of the Morpholino guanosine subunit for high-quality oligo production,15 as well as the creation of Vivo-Morpholino, a class of intracellularly penetrable Morpholino oligos for in vivo antisense studies.16
Yongfu Li, Ph.D., obtained his doctorate from the University of Vienna, Austria. After doing postdoctoral research work at the University of Cambridge, U.K., Li joined AVI Biopharma, Inc., USA (now Sarepta Therapeutics, Inc.). Four years later, he became head chemist at Gene Tools, LLC, and has been serving in that role ever since. For nearly 30 years, Li has gained extensive experience guiding structural modifications and Morpholino oligo functionalization. His most remarkable contributions to Morpholino chemistry have included the invention of double-protection of the Morpholino guanosine subunit for high-quality oligo production,15 as well as the creation of Vivo-Morpholino, a class of intracellularly penetrable Morpholino oligos for in vivo antisense studies.16
